Why Does Methane Have A Tetrahedral Shape In this video we explore the fascinating molecular geometry of methane CH using the Valence Shell Electron Pair Repulsion VSEPR Theory What You ll L
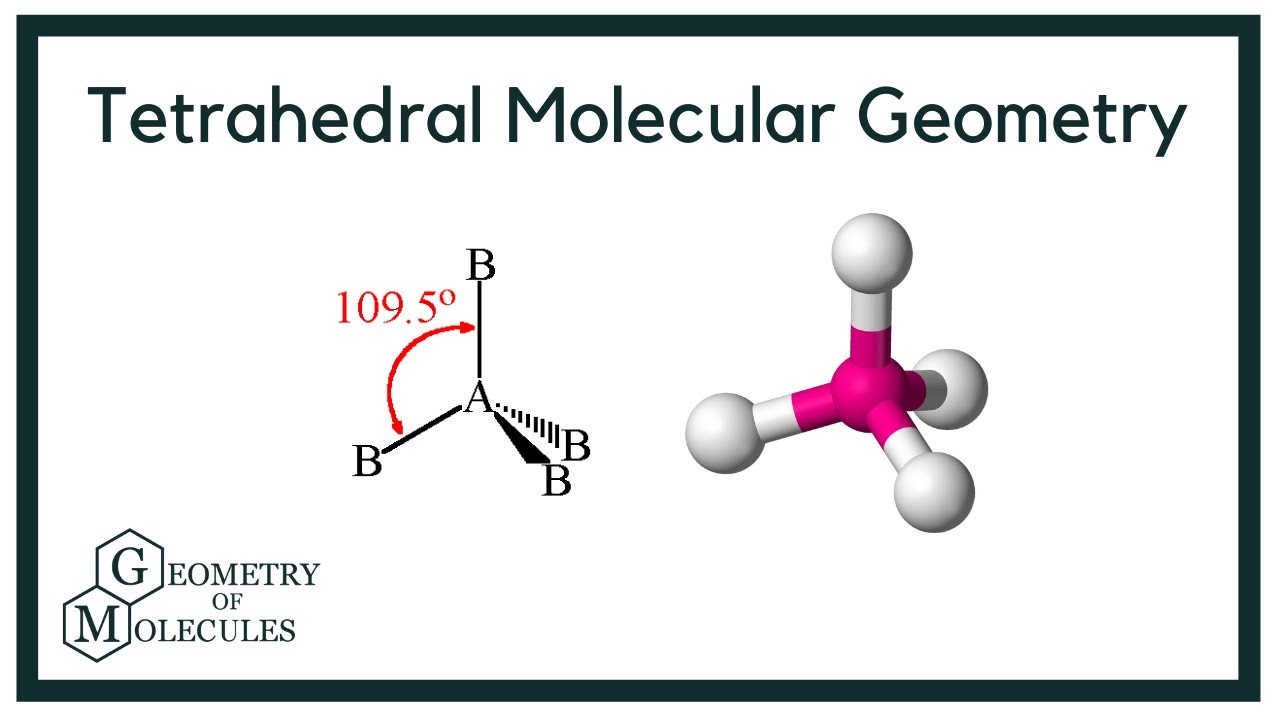
Methane and other perfectly symmetrical tetrahedral molecules belong to point group Td but most tetrahedral molecules have lower symmetry Tetrahedral molecules can be chiral The bond The tetrahedral shape in methane is determined by the repulsion between the four pairs of bonding electrons around the carbon atom In methane CH4 the central carbon atom is
Why Does Methane Have A Tetrahedral Shape
 Why Does Methane Have A Tetrahedral Shape
Why Does Methane Have A Tetrahedral Shape
https://i.ytimg.com/vi/OX6sKDh68rs/maxresdefault.jpg
Mar 13 2021 0183 32 Generally the lone pairs in the molecule distort the shape of the molecule which changes the molecule s bond angles But as there are no lone pairs of electrons in this
Templates are pre-designed documents or files that can be utilized for different functions. They can save effort and time by supplying a ready-made format and design for developing various kinds of material. Templates can be used for individual or expert projects, such as resumes, invitations, flyers, newsletters, reports, presentations, and more.
Why Does Methane Have A Tetrahedral Shape
Alkanes And Their Properties Cuestionario

Names Of Straight Chain Alkanes Diagram Quizlet

Chemistry Glossary Search Results For hybridization

Tetrahedral Examples
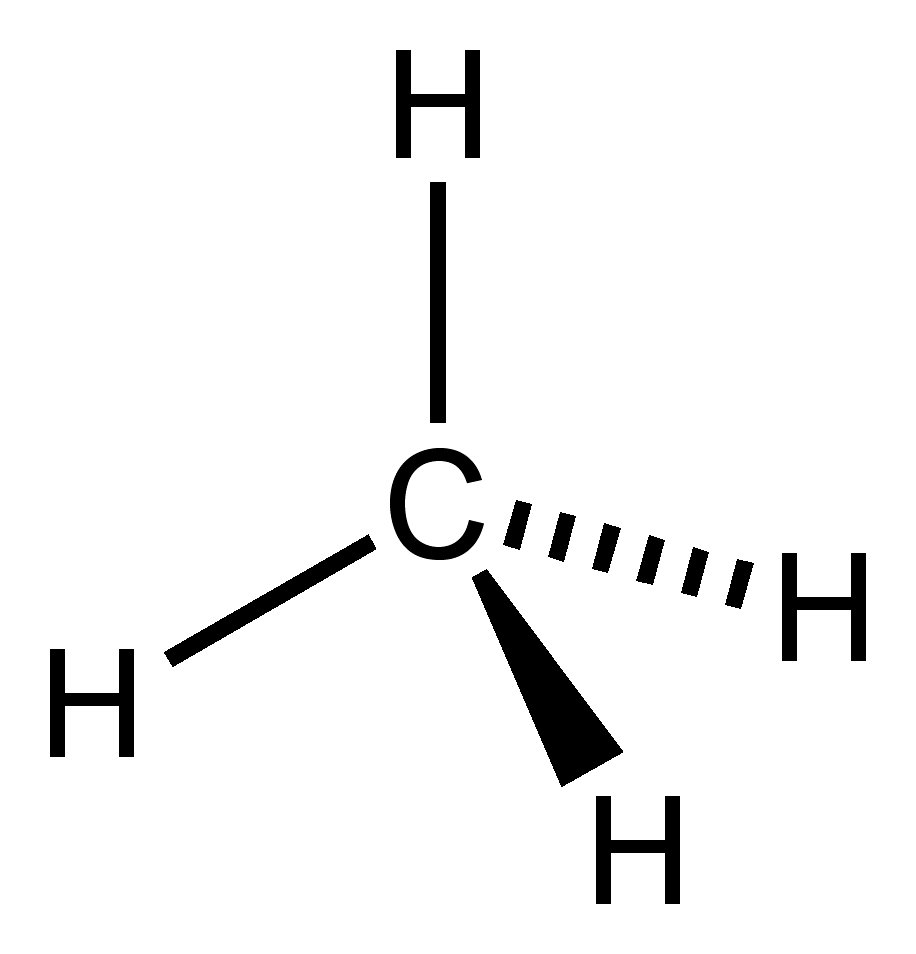
Methane Formula Structural And Chemical Formula Of Methane
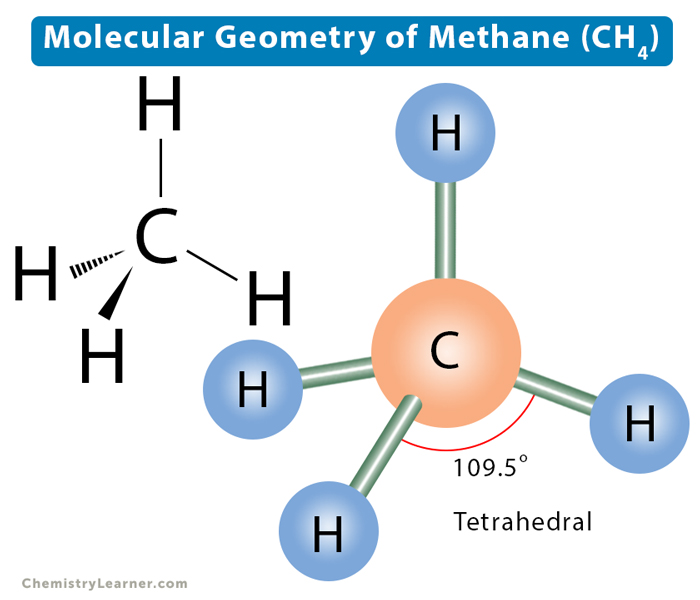
Molecular Geometry Lewis Structure Bond Angle Of Methane

https://chem.libretexts.org › Bookshelves › General
We can therefore predict the CH 3 N portion of the molecule to be roughly tetrahedral similar to methane The nitrogen atom is connected to one carbon by a single bond and to the other

https://topblogtenz.com
Oct 11 2023 0183 32 The Methane CH4 lewis structure is made up of one carbon atom that is situated at the middle position and four hydrogens atoms that are at the surrounding position CH4
https://chem.libretexts.org › Bookshelves › Organic
The formation of sp 3 hybrid orbitals successfully explains the tetrahedral structure of methane and the equivalency of the the four C H bonds What remains is an explanation of why the sp 3
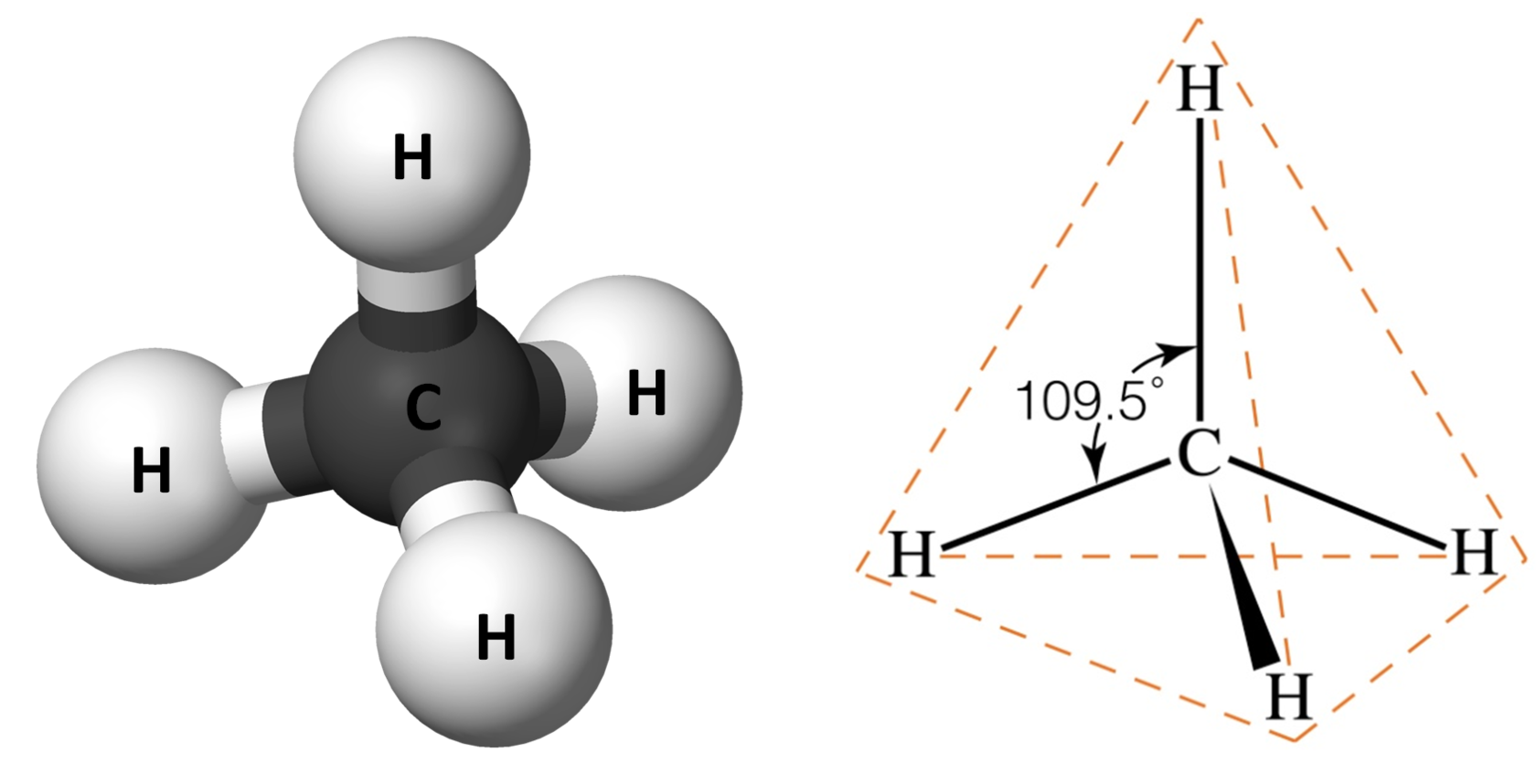
https://chem.libretexts.org › › Bonding_in_Methane
Jan 23 2023 0183 32 The shape of methane When sp 3 orbitals are formed they arrange themselves so that they are as far apart as possible That is a tetrahedral arrangement with an angle of
https://chemguide.co.uk › basicorg › bonding › methane.html
The shape of methane When sp 3 orbitals are formed they arrange themselves so that they are as far apart as possible That is a tetrahedral arrangement with an angle of 109 5 176 Nothing
Jun 3 2024 0183 32 Methane CH4 has a tetrahedral structure because it has four electron pairs around the central carbon atom These electron pairs repel each other leading to a shape that The H C H bond angle in methane is the tetrahedral angle 109 5 176 This angle is obtained when all four pairs of outer electrons repel each other equally The bond angles in ammonia and in
Jan 21 2025 0183 32 If there are four atoms attached to these electron groups then the molecular structure is also tetrahedral Methane CH 4 is an example Figure PageIndex 5